The Latest on the COVID-19 Vaccine – What Nurses Need to Know
Updated 12/21/2020
- President-Elect Joe Biden received the first dose of the Pfizer vaccine – the vaccine was administered live on television on Monday, December 21st.
- A female Tennessee nurse manager passed out after receiving the Pfizer vaccine – the situation was captured on camera, but according to both the nurse herself and the hospital that administered the vaccine, her fainting was not linked directly to receiving the vaccine. The nurse has an underlying condition in which her vagal response is overactive, which can cause her to have syncopal episodes and faint. “I had a syncopal episode. I have a history of having an overactive vagal response, and so with that if I have pain from anything — hangnail or if I stub my toe — I can just pass out,” the nurse, Tiffany Dover, told a news station.
- An empty vaccine was administered on camera – Meanwhile, in El Paso, TX,administered the vaccine to several nurses on camera, but when supposedly injecting the second nurse, the syringe appeared empty on camera, and the plunger was already depressed. After a news station reported on the apparent error, the University of Medical Center released a statement noting that the nurse had been re-vaccinated, just in case, as no adverse reactions are expected with a second vaccination. The incident serves to demonstrate both how the vaccine rollout can be challenging at a widespread scale and as a nurse, to always draw up your own medicine and follow all safety protocols before administering an injection.
- Allergic reactions to the vaccine – So far, a total of 6 allergic reactions to the Pfizer Biotech vaccine have been reported in total among the US that the CDC is reviewing. An Alaskan health worker had “probable” allergic reactions after receiving the first dose of the Pfizer vaccine, which included symptoms such as tongue swelling and trouble breathing. A healthcare worker in Illinois also had an allergic reaction, but after review, the health system the worker was vaccinated at resumed administering vaccines. According to Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, it’s thought that a common chemical used in cosmetics and household cleaners, polyethylene glycol, may be the culprit for the allergic reactions.
- The CDC recommends those with severe allergic reactions to not get the vaccine – If you have had a severe allergic reaction to any ingredient in the COVID-19 vaccine, you should not get the vaccine. If you have a history of severe allergic reactions that aren’t related to vaccines or injectable medications, however—such as allergies to food, pets, venom, environmental, or latex—you may still get vaccinated. People with a history of allergies to some medications taken by mouth may also still get vaccinated. Talk to your doctor about your own health history.
- The second COVID-19 vaccine, by Moderna, was approved for emergency use – the vaccine began arriving in states across the country on Monday.
- What are the differences between Pfizer and Moderna COVID vaccines? Here are the key differences and similarities between the vaccines.
- Some groups should not receive the vaccine at this time – As the vaccine rollout continues among states, there are some groups of people who may need to pass on the vaccine for now until we have more data in these specific populations. If you are among the following populations, talk to your own doctor about if and when you should get the vaccine:
- Young children (under the age of 18)
- Pregnant people
- People with weakened immune systems
- People who have or had COVID-19 already
- Anyone with allergies to any of the specific ingredients in the vaccine
Join The Conversation!
Join Nurse Alice Benjamin, APRN, MSN, ACNS-BC, FNP in a LIVE discussion about COVID-19 vaccine updates that matter most to nurses. This event takes place on 12/22/2020 at 6pm PST/9pm EST on @nurse_org Facebook and Instagram LIVE. Click here to set a reminder and join the conversation.
Updated December 14, 2020
The first phase of COVID-19 vaccinations began today Monday, December 14th, following the FDA’s emergency authorization of the Pfizer-BioNTech COVID-19 vaccine on Dec. 11. There is a lot that is still unclear about the vaccine process, such as who exactly will receive it, but here’s what we know so far:
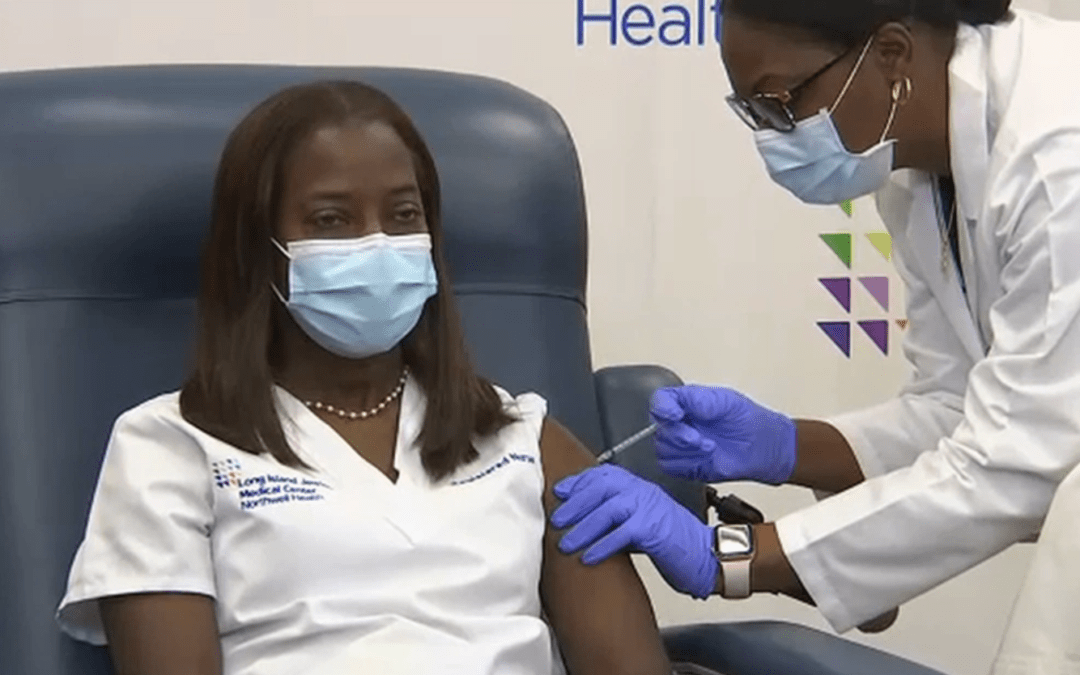
- ICU Nurse, Sandra Lindsay, of Long Island Jewish Medical Center in Queens, New York City, was one of the first nurses in the US to receive the Pfizer-BioNTech’s COVID-19 vaccine. The vaccine was administered on video.
- The first shipments of the vaccine have departed the Kalamazoo, MI Pfizer facility, heading out to bring the vaccine to all 50 states. The boxes of vaccines were carefully packed with both a GPS and thermal sensor, as the vaccine has to be kept at -94 Fahrenheit in order to keep the mRNA in the vaccine intact. 2.9 doses of the vaccine are set to be distributed this week, according to the New York Times.
- The vaccines will be distributed at a state level, so it’s up to each state to figure out who gets the vaccine, how, and where. In general, states are following CDC recommendations on vaccine administration. The CDC recommended that healthcare workers at high risk for exposure and long-term care residents receive the vaccine first.
- The specifics of the vaccine administration will be a challenge that state public health officials will be faced with this week; there is no centralized framework for funding, vaccine administration sites or staffing, or protocol for tracking who has received the vaccine and who is due for their second shot. (The Pfizer vaccine is given in 2 doses, 3 weeks apart. That means you have to receive 2 separate injections, 3 weeks apart, in order for the vaccine to be effective.)
- If you are a person who has been identified to receive the vaccine in the first phase, but you are pregnant or lactating, it is important for you to realize that you do have a right to receive the vaccine. While some facilities and organizations have, in the past, tried to restrict pregnant and breastfeeding individuals from receiving vaccines, the decision should be between you and your healthcare provider. Pregnant people typically are not included in vaccine or drug trials, which means there is officially very little data on the safety of any vaccine or medication during pregnancy. In the U.S., the ACOG has issued a formal recommendation that pregnant and lactating individuals not be excluded from the vaccine distribution. In Britain, however, the opposite approach was taken: they recommend that pregnant people do not get the vaccine and that pregnancy be avoided for the first two years after the second dose is given.
- The only people who should not get the vaccine as of now are people who have severe allergic reactions to vaccines in the past, or those who have had reactions to any of the ingredients in the vaccine.
- According to the FDA, the most common reported side effects of the Pfizer vaccine are: pain at the injection site, tiredness, headache, muscle pain, chills, joint pain, and fever. The NYT does note that even the mild side effects could cause healthcare professionals to miss a day or two of work, so the vaccine administration could pose additional challenges.
Because vaccine distribution is a state-led approach, the best place to check if you are eligible for a vaccine would be with your state health department. If you are a medical professional, your licensing bureau may also have some direction.
Updated December 11, 2020
The Pfizer-BioNTech COVID-19 vaccine was approved by the US Food and Drug Administration (FDA) for emergency use on Friday. Those at the highest risk for infection, including nurses, healthcare workers, and the elderly, will be the first in the US to have access to the first doses of the vaccine. It offers 95% protection against COVID-19.
“The FDA’s authorization for emergency use of the first COVID-19 vaccine is a significant milestone in battling this devastating pandemic that has affected so many families in the United States and around the world,” said FDA Commissioner Stephen M. Hahn, M.D.
According to President Trump, vaccinations will begin in less than 24 hours.
Updated December 3, 2020
- As of December 3, only one day after the U.S. recorded its single highest day of hospitalizations––surpassing 100,000––and deaths since the epidemic started–3,157 on Wednesday–a number higher than even the worst day in the spring, the U.S. is still waiting on an official vaccine approval.
- The U.K. authorized the Pfizer/BioNTech vaccine yesterday, beating the U.S. to accepting the vaccine. The U.S delay is due to the fact that the British regulators did not analyze the vaccine data as carefully as the U.S.’s FDA is doing currently, says Dr. Anthony Fauci.
- We still don’t have a clear timeline of when the U.S. can expect a COVID-19 vaccine and an FDA agency isn’t meeting until Dec. 10 to even consider an emergency authorization of the Pfizer vaccine––if they do decide to authorize it, the FDA will still have the decide after authorization is granted. However, according to Operation Warp Speed documents, public health officials are aiming to start distributing initial vaccines as soon as December 15th, with 20 million doses expected to be available.
- Once a vaccine is approved, there’s also a matter of mistrust in the vaccine by the American people. A large number of people polled are still undecided if they would not get the COVID-19 vaccine and former U.S. Presidents Clinton, Obama and Bush have all declared publicly that they will get the vaccine, even allowing themselves to be filmed while being injected, in order to bolster public trust.
- State governors are also being prepped on how many vaccines they should expect for their states, a number that is based on their over-age-18 population. Vaccines are to be distributed to identified populations first, according to the CDC, which is expected to include healthcare workers. And while the federal government will be providing the vaccines to the states, the states are responsible for their own distribution, which some state officials say could pose a significant problem with caps on end-of-the-year spending looming and healthcare facilities already stretched to the limit.
Original article posted on 11/9/2020
Nurses and healthcare workers will be amongst the first group to have access to the COVID-19 vaccine and get it if they choose.
In a new update with NPR, the CDC has stated that when a COVID-19 vaccine becomes available, nurses and other healthcare workers will be the first to have access to it.
In the interview with NPR, Dr. José Romero, who works as the head of the committee that develops evidence-based immunization guidelines for the CDC, explained that nurses, providers and healthcare workers will get first access to the vaccine because of their exposure to patients and their vital presence to keep the healthcare system running. According to Dr. Romero, an approved vaccine could be available as early as December or January. “We anticipate having some vaccine for the high-risk individuals — health care providers — sometime in December or early January,” said Romero.
He continued, “It’s not just the doctors and nurses that are interacting with patients, but also the support personnel that help. It could include those persons that are delivering food, or maintenance people that could come in contact with them.”
According to the CDC website, the goal of the vaccine recommendation is to “decrease death and serious disease as much as possible.”
And while some may see the advancement of a COVID-19 vaccine as a reason to celebrate, it’s also a reason to ask one very important question: do nurses want to get the vaccine?
On November 9, 2020 Pfizer and BioNTech announced that their “vaccine candidate was found to be more than 90% effective in preventing COVID-19 in participants without evidence of prior SARS-CoV-2 infection in the first interim efficacy analysis.”
How Nurses Feel About a COVID-19 Vaccine
If this year has shown us anything, it’s that we all tend to have very differing opinions on a lot of issues that affect this country–including the coronavirus. The pandemic has brought to light many differing opinions, beliefs and fears about what the future would hold, including how exactly a vaccine should be handled.
We shared the news with our Instagram community of over 121,000 nurses and hundreds of nurses commented. Read the responses here.
The issue over mandatory vaccines for healthcare workers is not a new one. A 2009 article in the Journal of Pharmacy and Therapeutics outlined the specific challenges in addressing mandatory vaccines for healthcare workers, namely balancing the rights of the healthcare worker to choose vaccination with the rights of patients and the preservation of public health. Despite recognizing the challenge of mandating vaccines, the article ultimately concluded, however, that choosing an occupation that involves, at its core, doing all in your power to improve the patient’s health means choosing to be vaccinated. And although encouraging healthcare workers to get vaccines is the preferred route, the article noted that in cases of non-compliance, mandating it may be unavoidable.
As you may expect, anticipating a vaccine requirement is causing mixed reactions among nurses. Jenny Erwin, a an RN and Vice President of her local nurses’ union from Michigan, has already had COVID-19, so she hopes her natural immunities will be enough should a vaccine be mandated. However, the nurse is skeptical about a vaccine and its future role in life post-virus.
“I’m a pro-vaccine for the record,” she tells Nurse.org. “ it seems crazy to me, that we still don’t even know a lot about this virus and now we have a vaccine? I’m not a clinical researcher or anything even close. How can we develop a vaccine if we don’t even know how people become immune to it? Or why some people have no symptoms at all? I hate how political COVID has become because it deserves respect. I’ve seen a lot of people die, quickly, with this virus. The CDC, etc. has lost a lot of respect with the way they handled COVID from the frontline workers. I want to believe in the science of it, I’m just so skeptical about everything right now.”
Vaccine Choice
The inevitable approval of a COVID-19 vaccine also brings into light the question of vaccine choice for nurses and other healthcare workers.
- Should they have the right to decline a vaccine?
- Should a vaccine be mandatory based on what type of patient population you are working with?
- What if you have COVID-19 antibodies already?
Mandatory vaccines, in general, is a heated topic and for travel nurses in particular, the question has already arisen. Some travel nursing positions posted this fall include a caveat: “Flu vaccine-no declinations.” However, it is important to note that some travel nursing positions have also made that requirement in years past too during flu season, so it’s not all just because of the `rona. But regardless of the origin, once the precedent for mandatory vaccination-or-no-job is set, some nurses are wondering if they will have a choice at all.
One interesting look at the topic comes from Sara Guercio, a Registered Nurse and law school graduate. Her article takes a deep dive into the legality of mandating vaccines and in short: there are no clear answers.
“As a nurse, with family and friends on the front lines of this epidemic, I want the maximum amount of protection for public health reasons; if that is a mandatory vaccine, I am all for it,” she writes. “But, as a nurse and an American woman, I am particularly concerned with privacy rights and the importance of the right to have a medical choice; I wonder and worry what precedent such a vaccination requirement would set.”
American Nurse’s Association’s Stance
If you’re wondering what the official nursing organizations have to say about it all, the American Nurses’ Association’s (ANA) stance on nurse immunizations is clear: vaccinate unless there is a medical exception. You can read their full policy, but the organization notes that they do not support vaccine exemptions based on “philosophical or religious exemptions” and they note that anyone who cannot receive a vaccine due to medical reasons “may be required to adopt measures or practices in the workplace to reduce the chance of disease transmission.”
** Nurse.org reached out to the CDC for further comment on this subject and will update the article once a response is received.
** Originally posted at Nurse.org


Recent Comments